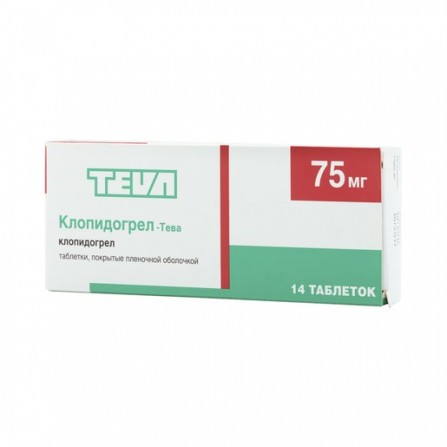
Clopidogrel Teva 75mg N14 coated pills
Condition: New product
1000 Items
Rating:
Be the first to write a review!

More info
Active ingredients
Clopidogrel
Release form
Pills
Composition
1 tablet contains: Active ingredient: clopidogrel 75 mg.
Pharmacological effect
Platelet aggregation inhibitor. Selectively inhibits the binding of adenosine diphosphate (ADP) to platelet receptors and the activation of GPIIb / IIIa complex, thus inhibiting platelet aggregation. Also inhibits platelet aggregation caused by other agonists, by blocking the increase in platelet activity by released ADP. Does not affect the activity of PDE. Clopidogrel irreversibly alters the ADP receptors on platelets, so platelets remain non-functional throughout "life", and restoration of normal function occurs as they renew (approximately 7 days).
Pharmacokinetics
After oral administration in a dose of 75 mg, clopidogrel is rapidly absorbed from the gastrointestinal tract. However, plasma concentration increases slightly and after 2 hours after administration does not reach a level that can be determined (0.025 mcg / l). Intensively metabolized in the liver. The main metabolite is an inactive carboxylic acid derivative and constitutes about 85% of the original substance circulating in the plasma. Cmax of this metabolite in the blood plasma after repeated doses of clopidogrel is about 3 mg / l and is observed approximately 1 h after administration. The pharmacokinetics of the main metabolite is characterized by a linear dependence in the dose range of clopidogrel 50-150 mg. Clopidogrel and the main metabolite irreversibly bind to plasma proteins in vitro (98% and 94%, respectively). This bond remains unsaturated in vitro over a wide range of concentrations. After ingestion of 14C-labeled clopidogrel, about 50% of the accepted dose is excreted in the urine and approximately 46% in the feces within 120 hours. The T1 / 2 of the main metabolite is 8 hours. Compared to healthy young volunteers, the concentration of the main metabolite in plasma is significantly higher elderly patients (75 years of age and older), with no changes in platelet aggregation and bleeding time. In severe kidney diseases (CK 5–15 ml / min), the concentration of the main metabolite in the blood plasma is lower than in case of moderately severe kidney diseases (CK 30–60 ml / min) and in healthy volunteers.Although the inhibitory effect on ADP-induced platelet aggregation was reduced compared with that in healthy volunteers, the bleeding time increased to the same extent as in healthy volunteers.
Indications
Prevention of thrombotic complications in patients with myocardial infarction, ischemic stroke or occlusion of peripheral arteries. In combination with acetylsalicylic acid for the prevention of thrombotic complications in acute coronary syndrome: with ST segment elevation with the possibility of carrying out thrombolytic therapy without ST segment elevation (unstable stenocardia, myocardial infarction without Q wave), including in patients undergoing stenting.; Prevention of thrombotic and thromboembolic complications, including stroke, in atrial fibrillation (atrial fibrillation) in the presence of at least one risk factor for the development of vascular complications, inability to receive indirect anticoagulants and low risk of bleeding (in combination with acetylsalicylic acid).
Contraindications
Acute bleeding (including with peptic ulcer or intracranial hemorrhage), severe liver failure, pregnancy, lactation (breastfeeding), children and adolescents under 18 years of age, increased sensitivity to clopidogrel.
Dosage and administration
Is ingested 1 time / day. The initial and maintenance dose is 75 mg / day. Loading dose - 300 mg / day. The regimen depends on the indications and the clinical situation.